In order to implement the ASEAN Cosmetics Agreement and the provisions of Circular No 2. Specific information about the updated content in the Annexes of the ASEAN Cosmetics Agreement, Vietnam FDA updated the Ingredients lists as follows and it will take effect on 11.05.2023.
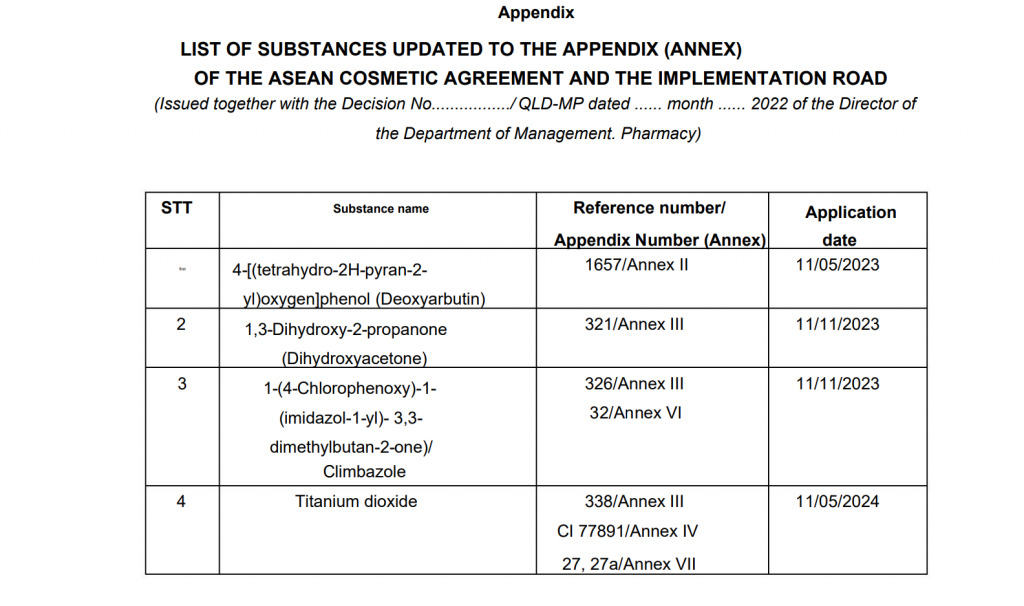
– Appendix II. List of substances that are not allowed to be used in cosmetic products;
– Appendix III. List of substances with regulations on concentration and content limits in cosmetic products;
– Appendix IV. Colorants are allowed to be used in cosmetics;
– Appendix VI. List of preservatives allowed to be used in cosmetic products;
– Departments of Health of provinces and centrally run cities
– Appendix VII. List of UV filters.